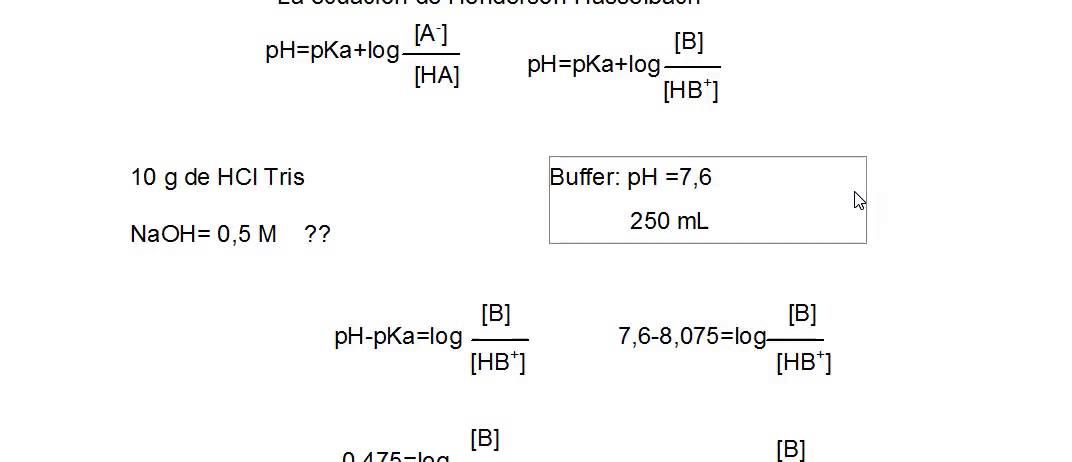 
Notice that the pH is greater than the solution of just the pure acid (4.752 as compared to 2.376). Since the log of 1 is zero, we have pH = 4.752Ĭomment: 1.8 x 10¯ 5 is a commonly-seen value for the K a of acetic acid. PK a = −log K a = −log 1.77 x 10¯ 5 = 4.752ģ) Next, we simply insert the appropriate values into the H-H equation: Tables of K a values are also widespread on the Internet. Even in this era of fairly easy Internet access, try one of the appendices of your textbook. If the problem provides the K a, you must convert it to the pK a (see below).Ĭomment: be aware, your teacher may create a test question where you must look up the K a. Often, the problem will provide the pK a. What is its pH?ġ) To solve the above example, we must know the pK a of acetic acid. Knowledge of the inner workings of the H-H Equation will be to your chemical benefit.Įxample #1: A buffer is prepared containing 1.00 M acetic acid and 1.00 M sodium acetate. The ChemTeam recommends you become familiar with H-H calculations, then return to the Deep Dive and give it a careful read. 
At the end, he details a case where the approximation fails and an exact calculation must be made. In his document, he goes into detail concerning the approximations that are used to develop the Henderson-Hasselbalch Equation and why those approximations are valid (hint: the 5% rule). The third set of problems (#21 to 30) has more examples of this type.Ī colleague has written a Deep Dive into the first three examples solved just below. There will be an unknown in the log portion of the Henderson-Hasselbalch. Note: the bonus problem at the end of the file involves having to calculate how much of one of the buffer components is consumed and how much of the other is produced. Many Q&A forums still lack the ability to make a more typeset-appearing H-H Equation. Is often the way you see it written on the Internet. to the Henderson-Hasselbalch Equation Return to the Acid Base menuĪs a reminder, here is the Henderson-Hasselbalch Equation: ChemTeam: Buffers and the Henderson-Hasselbalch Equation: Fifteen Examples Buffers and the Henderson-Hasselbalch Equation:įifteen Examples Buffer Problems 1-10 Buffer Problems 11-20 Buffer Problems 21-30 Buffer Problems 31-40 Intro.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
